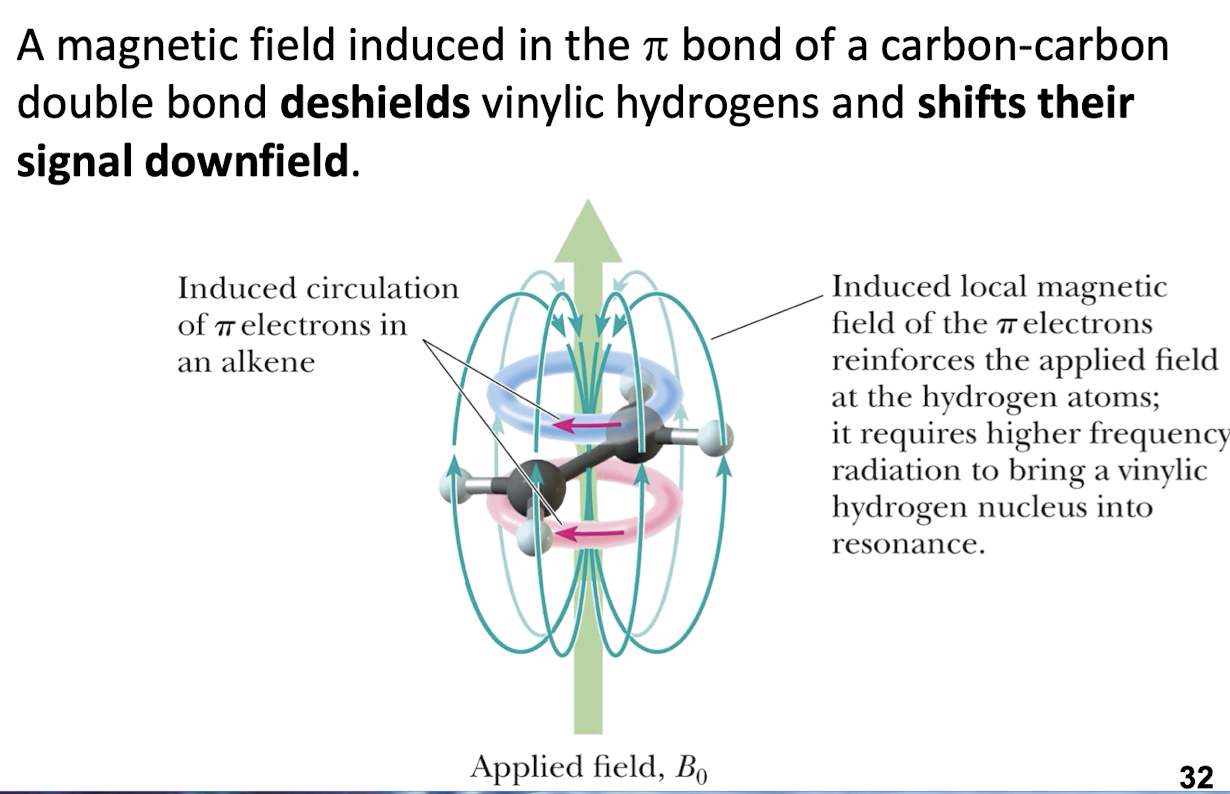
Ansiotropic Effect
#Chemistry
Refers to the idea that protons attached to aromatic rings are more deshielded than expected. This occurs because $\displaystyle \vec{B}$ is induced in the same direction as the incident $\displaystyle \vec{B}_{0}$. The $\displaystyle \vec{B}$ occurs like below
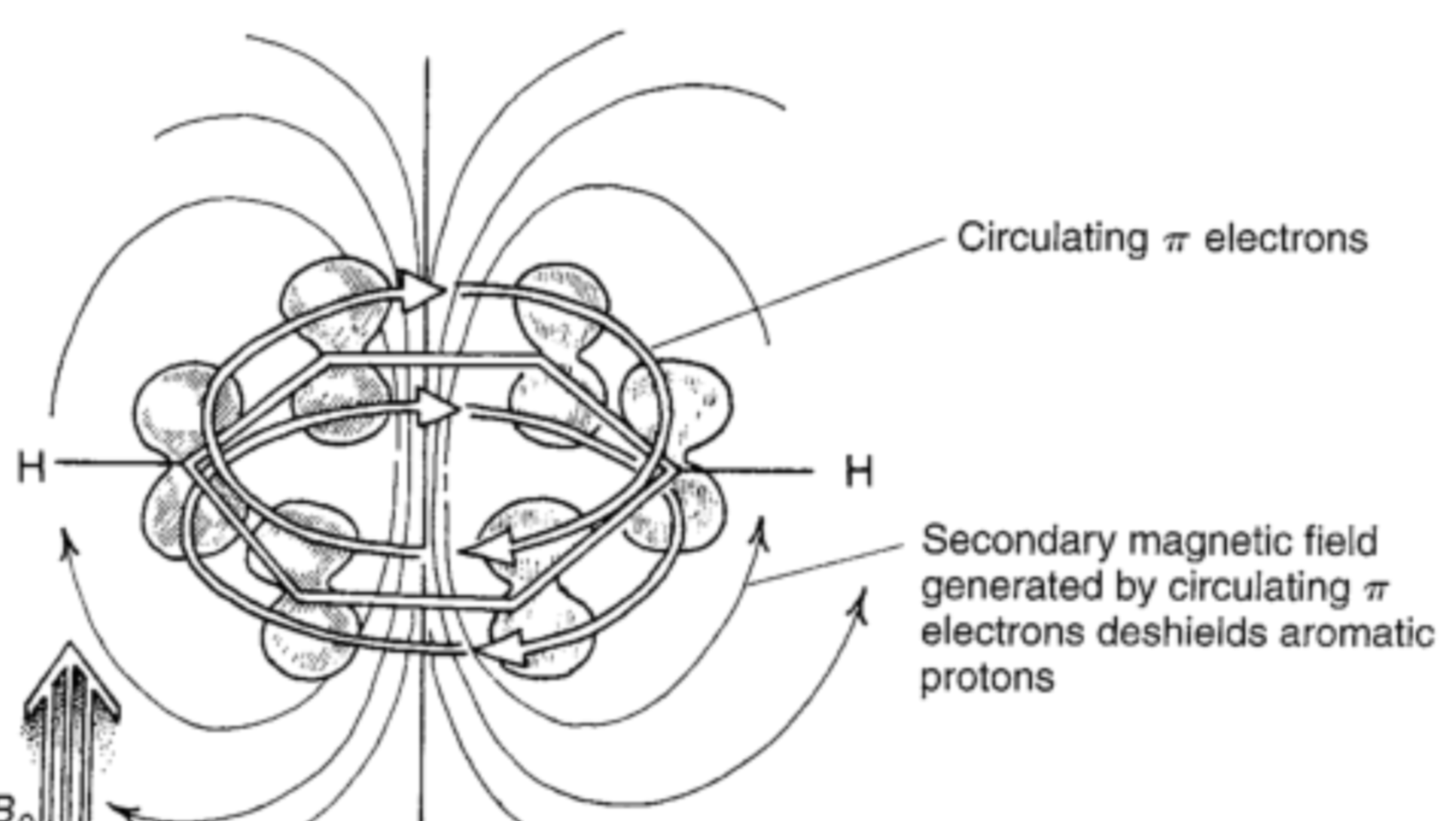
In this diagram, the current is conventional current. When the aromatic compound is first placed in the magnetic field, magnetic flux increases, which then kick starts the electrons in a counter clockwise flow in this diagram. Due to the Lorentz force, the electrons continue circulating and thus producing the magnetic field which contributes to
Alkyne
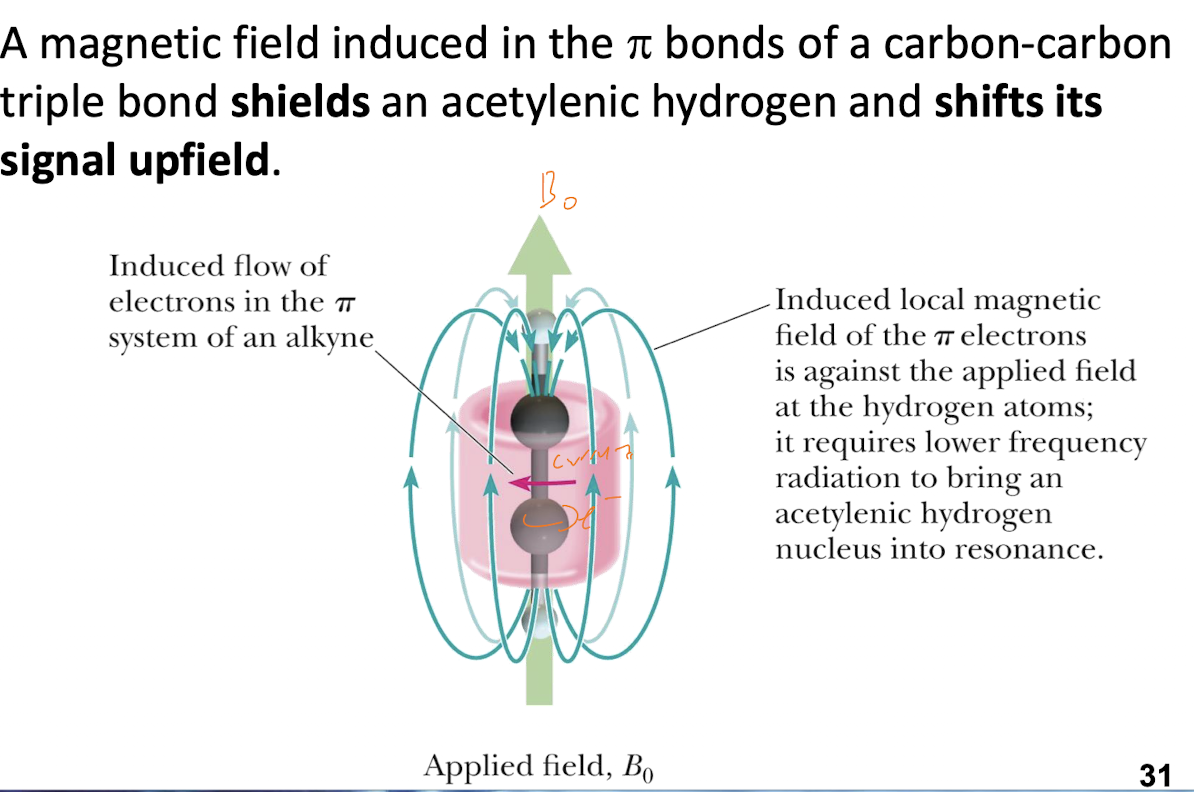
Alkene