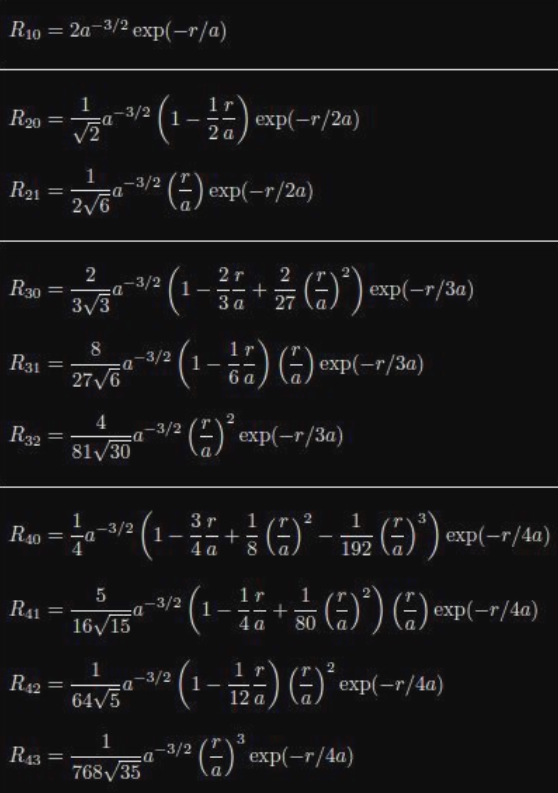Hydrogen Atom
#Physics
Topics
$\displaystyle V(r)=-\frac{e^{2}}{4\pi {\varepsilon}_{0}} \frac{1}{r}$
- This is the potential for an electron surrounding a hydrogen atom
$\displaystyle E_{n}=-\left[ \frac{m_{e}}{2\hbar ^{2}}\left( \frac{e^{2}}{4\pi {\varepsilon}{0}} \right)^{2} \right] \frac{1}{n^{2}}= \frac{E{1}}{n^{2}}, ,n\in \mathbb{Z}^{+}$
- Allowed energy levels of a
$\displaystyle E_{1}=\left[ \frac{m_{e}}{2\hbar ^{2}}\left( \frac{e^{2}}{4\pi {\varepsilon}_{0}} \right)^{2} \right]=-13.6\mathrm{,eV}$
- Equivalent to binding energy of hydrogen atom to electron/ionization energy
$\displaystyle \psi_{100}(r,\theta,\phi)=\frac{1}{\sqrt{ \pi a^{3} }}e^{-r /a}$
- Ground state of hydrogen
$\displaystyle \psi_{nlm}(r,\theta,\phi)=R_{nl}(r)Y_{l}^{m}(\theta,\phi)$
- Wave function of a hydrogen atom for quantum numbers $\displaystyle n$, $\displaystyle l$, and , $\displaystyle m$
- $\displaystyle R_{nl}$ is given in the equation below and is the radial wave function for the hydrogen atom
- $\displaystyle Y_{l}^{m}(\theta, \phi)$ is the Spherical Harmonics
$\displaystyle R_{nl}(r)=\sqrt{ \left( \frac{2}{na_{0}} \right)^{3} \frac{(n-l-1)!}{2n(n+l)!} }e^{-r /na_{0}}\left( \frac{2r}{na_{0}} \right)^{l}\left[ L_{n-l-1}^{2l+1}\left( \frac{2r}{na_{0}} \right) \right]$
- The first term under the square root is a normalization factor
- $\displaystyle a_{0}$ is the Bohr radius
- $\displaystyle L_{n-l-1}^{2l+1}$ is the associated Laguerre polynomial
$\displaystyle R_{nl}(r)$ Values