IR Spectroscopy
#Chemistry
Infrared light goes from 400 to 4000 $\displaystyle \mathrm{cm^{-1}}$, or
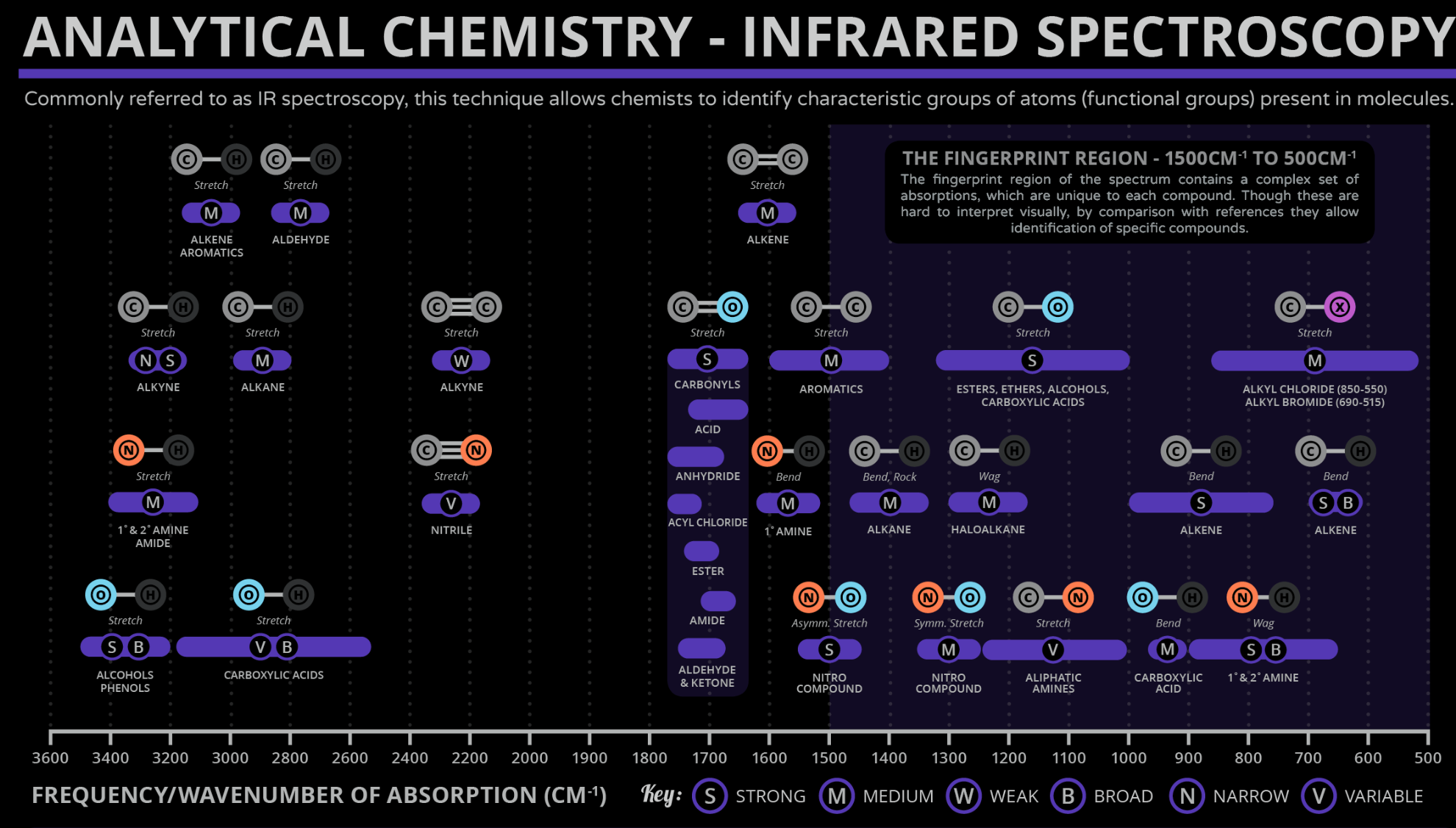
Zones of IR Spectroscopy
- The Polar $\displaystyle \ce{H}$'s
- The $\displaystyle \ce{C-H}$'s
- The Triple Bonds
- The Carbonyls
- The Double Bonds
| Bond | Stretching Frequency ($\displaystyle \mathrm{cm^{-1}}$) | Intensity and Shape | Zone |
|---|---|---|---|
| Alcohol $\displaystyle \ce{O-H}$ | 3650-3200 | Usually strong and broad | 1 |
| Alkyne $\displaystyle \ce{C\equiv H}$ | 3340-3250 | Usually strong and sharp | 1 |
| Amine or amide $\displaystyle \ce{N-H}$ | 3500-3200 | Medium; often broad | 1 |
| Aryl or vinyl $\displaystyle sp^{2},\ce{C-H}$ | 3100-3000 | Variable | 2 |
| Alkyl $\displaystyle sp^{3},\ce{C-H}$ | 2960-2850 | Variable | 2 |
| Aldehyde $\displaystyle \ce{C-H}$ | ~2900, ~2700 | Medium; two peaks | 2 |
| Carboxylic acid $\displaystyle \ce{O-H}$ | 3000-2500 | Usually strong; very broad | 2 |
| Alkyne $\displaystyle \ce{C\equiv C}$ | 2260-2000 | Variable and sharp | 3 |
| Nitrile $\displaystyle \ce{C\equiv N}$ | 2260-2220 | Variable and sharp | 3 |
| Ketone $\displaystyle \ce{C=O}$ | 1750-1705 | Strong | 4 |
| Ester $\displaystyle \ce{C=O}$ | 1750-1735 | Strong | 4 |
| Aldehyde $\displaystyle \ce{C=O}$ | 1740-1720 | Strong | 4 |
| Carboxylic acid $\displaystyle \ce{C=O}$ | 1725-1700 | Strong | 4 |
| Amide $\displaystyle \ce{C=O}$ | 1690-1650 | Strong | 4 |
| Alkene $\displaystyle \ce{C=C}$ | 1680-1620 | Variable | 5 |
| Benzene $\displaystyle \ce{C=C}$ | ~1600; ~1500-1450 | Variable; 1600 often two peaks | 5 |
| Below 1400 $\displaystyle \mathrm{cm^{-1}}$ is the fingerprint region, which mostly contains bending vibrations and unfortunately a lot of noise |
IR Spectroscopy Chart
$\displaystyle \bar{\omega}=\frac{1}{2\pi c}\sqrt{ \frac{k}{\mu} }$
- The stretching vibrational frequency of light
- $\displaystyle c$ is the speed of light
- $\displaystyle k$ is the spring constant
- $\displaystyle \mu$ is the reduced mass