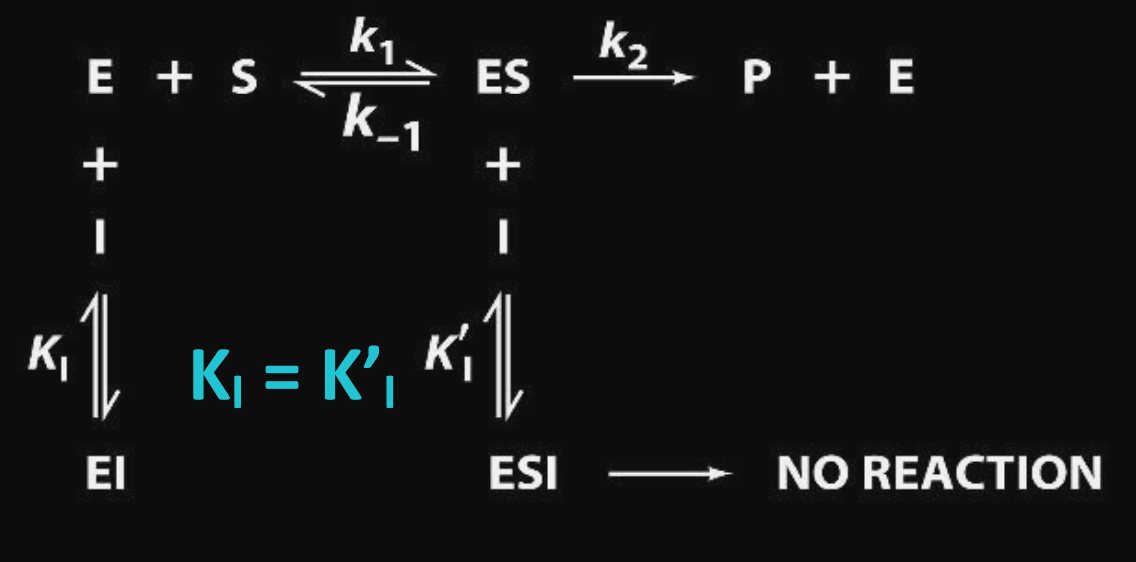
Mixed Inhibition
#Biochemistry
Inhibitor binds allosterically to either the enzyme or ES complex. We define $\displaystyle K_{I}$ to be the dissociation constant for the free $\displaystyle \ce{EI}$ complex (equilibrium constant for converting $\displaystyle \ce{EI->E + I}$) and $\displaystyle K_{I}'$ to be dissociation constant for the $\displaystyle \ce{ESI}$ complex
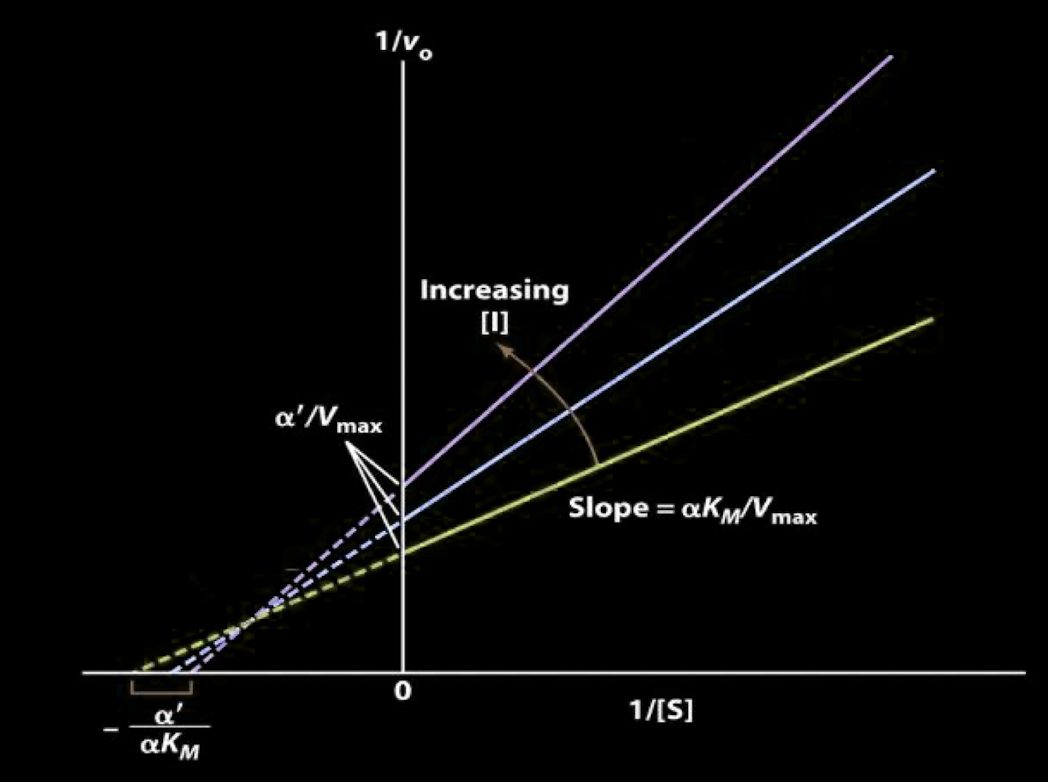
- $\displaystyle K_{m}$ increased if $\displaystyle K_{I}<K_{I}'$ where $\displaystyle K_{m}$ is the Michaelis constant
- If the inhibitor binds more strongly to E than it does to ES, binding affinity of E to S decreases, so $\displaystyle K_{m}$ increases
- This is kind of analogous to competitive inhibition since for lower relative $\displaystyle K_{I}$, we'll have less free $\displaystyle E$, which is kind of how the competitive inhibitor takes away free $\displaystyle E$. Competitive inhibition also increases $\displaystyle Km$
- $\displaystyle K_{m}$ decreased if $\displaystyle K_{I}>K_{I}'$
- If the inhibitor binds more strongly to ES than it does to E, binding affinity of E to S increases, so $\displaystyle K_{m}$ decreases
- $\displaystyle v_{\text{max}}$ decreased due to general inhibition
- Desmos Graph of Lineweaver-Burk Plot for Mixed Inhibition
Lineweaver-Burk Plot