Spectrum Energy
#Physics
$\displaystyle E_{\gamma}=E_{i}-E_{f}=-13.6\mathrm{eV}\left( \frac{1}{n_{i}^{2}}-\frac{1}{n_{f}^{2}} \right)$
$\displaystyle \frac{1}{\lambda}=\mathcal{R}\left( \frac{1}{n_{f}^{2}}-\frac{1}{n_{i}^{2}} \right)$
- $\displaystyle \mathcal{R}$ is the Rydberg constant
Transitions
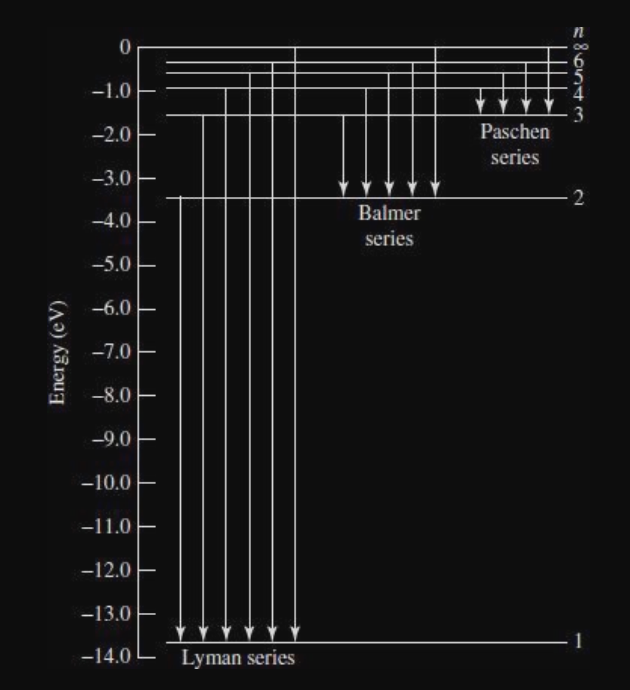
- Lyman series corresponds to ultraviolet
- Balmer series corresponds to visible
- Paschen corresponds to infrared